In scientific analysis, especially in fields such as drug development, cancer studies, and genetic testing, cell lines play a vital role. These are groups of cells that are grown in labs and used to study different biological processes. However, numerous times, cell lines get contaminated or mixed up without anyone realizing it. This can lead to incorrect results, wasted time, and financial loss. To prevent such errors, experts utilize a specialized test known as cell line authentication. This test helps prove whether the cells being utilized are exactly what they are supposed to be. It checks the unique DNA of the cell line and compares it with known references to ensure authenticity. By performing this test, researchers can be confident in their work, publish accurate findings, and meet international research standards. It is a simple but important step in keeping the quality of the cell line of interest.
Why is It Necessary?
- Studies indicate that 15–30% of cell lines used in research are either cross-contaminated or misidentified.
- This has led to flawed experiments, wasted funding, and even retractions of published papers.
- Mislabeled or contaminated cell lines can delay drug development or even lead to quality conclusions.
Here’s why the cell line authentication test in India is gaining significance:
- Accurate & Reliable Results – If the incorrect cell line is used, the whole analysis could be based on false assumptions. Correct and accurate identification guarantees that scientific data is valid and can be trusted.
- Saves Money and Time- Imagine spending years on a research project only to find out that the cell line used was wrong. Authentication saves time and prevents financial losses.
- Publication Requirement – Most reputed scientific journals and institutions now require a certificate of authentication of the cell line before accepting any paper.
- Research Integrity – Good science is built on precision and transparency. Authentication of the cell line upholds these values.
How Does It Work?
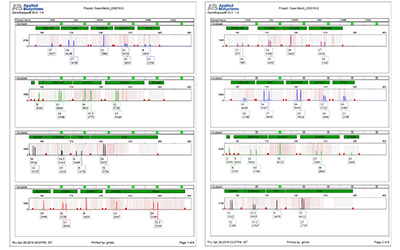
This test usually involves STR (Short Tandem Repeat) profiling — a genetic test method also used in human DNA fingerprinting. Following is the step-by-step explanation of the process:
- Sample Collection: The DNA testing lab gets the DNA from the cell line, either directly or as extracted DNA.
- DNA Extraction: If the DNA is not already extracted, the lab will perform DNA isolation from the cell sample.
- STR Analysis: Short Tandem Repeat markers — small repeated DNA segments — are analyzed using PCR (Polymerase Chain Reaction).
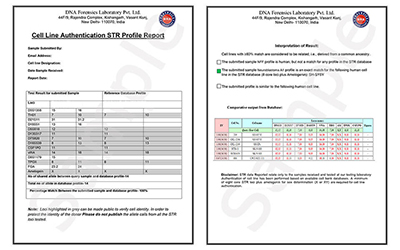
- Profile Matching: The test results are compared with standard cell line databases like ATCC, NIH, NCBI, or ICLAC.
- Report Generation: A detailed report is issued showing whether the sample is authenticated or contaminated, along with genetic match percentages.
Who Needs It?
Many industries and research sectors rely on experimental with biological cell lines. These include:
- Medical Research: Especially in cancer biology, genetics, and infectious diseases. Using the correct cell line is critical for the success of life-saving research.
- Pharmaceutical Companies: Before launching new drugs, pharmaceutical companies test them on specific cell lines.
- Biotech Companies: Startups and companies working on innovative biological research and product development may frequently need to establish their credibility with authenticated results.
- Academic Institutions: Universities and colleges involved in biological research projects are expected to use validated cell lines.
- Government & Private Laboratories: Government and private labs working on vaccines or disease models often rely on authenticated cell lines.
Commonly Misidentified Cell Lines
Over the years, numerous cell lines have been discovered to be misidentified. Some cases are:
- HeLa Cells: HeLa Cells frequently contaminate other cell cultures due to their aggressive growth.
- KB Cells: Believed to be oral cancer cells but later identified as HeLa derivatives.
- MCF-7: A widely used breast cancer line that has been mixed up with other cells in several cases.
These examples underline the importance of getting your cell lines authenticated before starting any serious research.
Benefits of Authentication of Cell Lines in India
India is rapidly becoming a hub for scientific analysis and innovation. Some benefits are-
- Availability of Cutting-Edge DNA Technologies
- Skilled and Qualified Specialists
- Cost-Effective Testing
- Accredited Labs (NABL and ISO-certified)
- Quick Sample Collection Across the Country
- Fast Reporting Time
Whether you’re a student, researcher, or biotech professional, getting your cell lines authenticated is now more accessible than ever.
Why DNA Forensics Laboratory?
DNA Forensics Laboratory Pvt. Ltd is among the most trusted companies for DNA testing. Let’s understand why we are the top choice for this test-
- Experienced and Trusted Lab: We are a well-established and experienced genetic testing company. We have been serving clients across India and globally with a variety of DNA testing services, including paternity tests, forensic tests, and cell line authentication DNA tests in India. With years of experience in human genetic testing, we bring the same level of accuracy and reliability to cell line services.
- Advanced STR Profiling Technology: This test is done through Short Tandem Repeat (STR) profiling, which checks the DNA fingerprint of the cell line. DNA Forensics Laboratory uses international standard methods and advanced STR profiling systems to ensure precise results. Their test follows the ATCC and NIH guidelines, which are accepted globally by researchers and journals.
- Fast and Accurate Results: We offer a quick turnaround time — most reports are delivered within 10-15 working days. Our detailed reports are clear, easy to understand, and include full genetic profiles along with comparisons to reference databases.
- Accredited and Certified: Our lab is NABL and ISO certified, which means our procedures meet international quality standards. This adds extra assurance that your results will be accurate, reliable, and accepted by major journals, institutions, and regulatory bodies.
- PAN-India Sample Collection: No matter where you reside in India, we can help. We have a wide network of sample collection centers across India and many other countries. You can easily schedule a pickup, courier your sample, or visit our nearby facility.
- Competitive Pricing: We offer high-quality authentication of the cell line at competitive costs, ensuring excellent value for money. Our pricing is designed to support students, researchers, universities, and biotech companies, making this essential service accessible without straining their budgets.
- Excellent Customer Support: Our customer service team is friendly and well-versed. We guide you through the entire process — from how to send your sample to understanding your report.
You can visit our lab or book your cell line authentication DNA test in India, call at +91 8010177771 or WhatsApp at +91 9266615552.